-
PDF
- Split View
-
Views
-
Cite
Cite
M Génard, N Bertin, C Borel, P Bussières, H Gautier, R Habib, M Léchaudel, A Lecomte, F Lescourret, P Lobit, B Quilot, Towards a virtual fruit focusing on quality: modelling features and potential uses, Journal of Experimental Botany, Volume 58, Issue 5, March 2007, Pages 917–928, https://doi.org/10.1093/jxb/erl287
Close - Share Icon Share
Abstract
The fruit is a hierarchically organized organ composed of cells from different tissues. Its quality, defined by traits such as fruit size and composition, is the result of a complex chain of biological processes. These processes involve exchanges (transpiration, respiration, photosynthesis, phloem and xylem fluxes, and ethylene emission) between the fruit and its environment (atmosphere or plant), tissue differentiation, and cell functioning (division, endoreduplication, expansion, metabolic transformations, and vacuolar storage). In order to progress in our understanding of quality development, it is necessary to analyse the fruit as a system, in which processes interact. In this case, a process-based modelling approach is particularly powerful. Such a modelling approach is proposed to develop a future ‘virtual fruit’ model. The value of a virtual fruit for agronomists and geneticists is also discussed.
Introduction
Fruit quality involves a set of traits such as fruit size, overall composition and taste, and proportion of edible tissue. The links between environmental control and quality traits have been extensively investigated (Ho, 1998; Bertin et al., 2000; Wu et al., 2002) and progress in the understanding of the genetic control of quality has been boosted by the emergence of molecular markers and mapping technology (Causse et al., 2002; Lecomte et al., 2004). However, these studies have limited explanatory power since they focus on the links between control variables and quality traits, without explicitly considering the underlying mechanisms. In order to progress in our understanding of quality build-up, it is necessary to analyse the fruit as a system characterized by interacting biological processes. For this purpose, the modelling features of a virtual fruit are presented here. Even though every process involved in fruit physiology cannot be integrated in a virtual fruit, a real degree of complexity is needed. Indeed, fruit has the characteristics of complex systems as defined by Wu and Marceau (2002). In particular, it exchanges energy and/or mass with its environment (atmosphere, plant) and it is composed of a large number of diverse components (different sugars and acids, tissues, etc) which interact with each other non-linearly.
This complexity has been investigated recently through the development of several models which describe the main processes involved in the build-up of fruit quality (Génard and Lescourret, 2004) and constitute a strong knowledge base for a virtual fruit project. The goal of this article is to propose a general system of organization for a virtual fruit, including the processes to be considered at different scales, and their control. The value of a virtual fruit for agronomists and geneticists is pointed out.
How should the fruit system be represented?
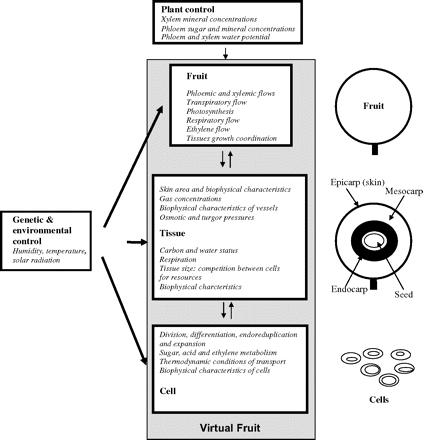
In order to unravel the complexity of the fruit system, it is proposed to analyse it following a hierarchical approach (Wu and David, 2002). The fruit is composed of one or several seeds, surrounded by a pericarp, itself composed of three tissues: the endocarp which may be lignified as in the case of stone fruits; the mesocarp which is composed of large cells constituting the main part of the fruit; and the epicarp (skin). This structure can be defined at three main levels. At the higher level, the fruit can be considered as an entity exchanging resources (minerals, sugars, and water) with the plant through the phloem and xylem vessels, and gases (water vapour, CO2, O2, and ethylene) with the atmosphere through the skin. At an intermediate level, the tissues can be treated as separate compartments in interaction and/or competition. Each of these tissues has specific characteristics in terms of ontogenesis, growth features, metabolism abilities, and construction costs (Valantin et al., 1999). At the lower level, the fruit is composed of cells subject to different processes such as cell division, cell differentiation, cell endoreduplication, cell expansion, metabolic transformations, and vacuolar storage. A schematic representation of the virtual fruit organization is presented in Fig. 1.
Schematic representation of the virtual fruit organization in levels and objects (on the right). For each level, constraints on the lower level are given in the lower part of the box and initiating conditions to the upper level are given in the upper part of the box. The plant, genetic, and environmental controls are also indicated.
Which level-to-level and process-to-process connections?
In a hierarchical approach, the relationship between two levels is asymmetric: a higher level exerts constraints on the level below it, whereas the lower level provides initiating conditions to the higher level (Fig. 1).
At the cellular level, cell differentiation governs the tissue's appearance, and the intensity of cell division and endoreduplication, which are important components of sink strength (Ho, 1996; Sugimoto-Shirasu and Roberts, 2003). The biophysical characteristics of cells govern tissue growth, while cell metabolism and vacuolar storage govern the tissue composition in sugars, acids, and secondary compounds.
At the tissue level, the biophysical characteristics of a tissue, its carbon and water status, its growth and maintenance respiration costs, and its size in terms of cell number drive the growth and metabolism at the cell level. The area and biophysical characteristics of skin and the gas concentrations in tissues control the gas exchanges of the fruit with the atmosphere. The phloem and xylem conductances and the osmotic and turgor pressures of tissues determine the water, minerals, and carbon exchanges with the plant. The upper level is the fruit, where the different fluxes (phloemic and xylemic fluxes, transpiration, respiration, etc.) drive the water and carbon status as well as gas concentrations in the fruit tissues. It is also at this level that the relative growth of tissues is co-ordinated.
In such a hierarchical system, information passes between the different levels. For instance, from the cell to the fruit level, the cell ploidy governs the cell sink strength and the cell division governs the cell number in the tissues. The cell number and cell sink strength govern the tissue sink strength which is an important factor of the phloemic flux into the fruit. Now from the fruit to the cell level, the phloemic flux drives the assimilate supply to the tissues. This supply is shared among the cells according to their individual sink strength, and the cells can divide, grow, and accumulate sugars and acids.
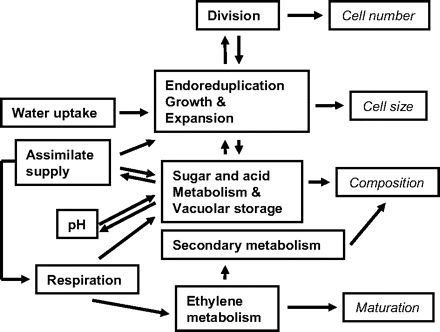
A complete consideration of feedback and interaction mechanisms also requires an accurate representation of the links among physiological processes at a given level. The first process to consider in the fruit life time is cell division. After a first phase of division, cell DNA endoreduplication boosts the cell sink strength. Cell expansion then depends on the flux of water and on metabolic transformations. The metabolic transformations, vacuolar storage, and ethylene metabolism, which determine the biochemical composition and maturation, depend on resources, pH, and other processes such as respiration. A schematic representation of the relationships among processes at the cell level is presented in Fig. 2. At the tissue level, the intensities of cellular processes and of their interrelationships vary according to the type of tissue. Tissue-specific properties such as mechanical properties depend on the interaction between tissue differentiation and cell density in the tissue. At the fruit level, it is important to preserve the respective proportions of the tissues throughout the season by applying laws of assimilate distribution among them. Strong relationships between phloem and xylem fluxes and transpiration exist (Huguet et al., 1998; Li et al., 2001; Kawabata et al., 2005), and a virtual fruit should be able to simulate these relationships.
Schematic representation of the relationships between processes at the cell level as considered in the virtual fruit, and links with cell number, size, composition, and maturation (italics). Division was assumed to be related to assimilate supply, water uptake, and other processes through its link with cell growth. The cell growth and expansion were dependent on water uptake, assimilate supply, DNA endoreduplication, and metabolism. The sugar and acid metabolism which determines fruit acidity and sweetness interacts with a number of processes such as assimilate supply and respiration. The ethylene metabolism which was related to fruit maturation and accumulation of secondary compounds was assumed to be controlled by respiration. In numerous cases, feedback loops are involved in the link between processes. Models are available for all the processes except for the secondary metabolism.
Which processes to consider and how?
Each process to be included in a single complex model must be described in a simple fashion. Following Tardieu (2003), one can assume that the network of gene regulation is co-ordinated in response to internal or external factors, giving way to meta-mechanisms; this allows a simplified mechanistic representation of the processes. Hereafter, existing process-based models which could be included in a virtual fruit will be reviewed.
Cell scale
Cell division, differentiation, size increase, metabolic transformations, and vacuolar storage are the essential processes to be considered at the cell scale. They determine important quality traits such as fruit size, sweetness, acidity, and nutritional quality.
Cell division:
Several comprehensive models of mitotic control have been proposed recently (Novac and Tyson, 1993; Gardner et al., 1998; Ciliberto and Tyson, 2000), but their complexity is too great to be integrated in a virtual fruit. A phenomenological population dynamic model was suggested for the analysis of cell multiplication in growing fruits (Bertin et al., 2003). This model involves a phase of exponential cell proliferative activity, followed by a progressive decrease of proliferative activity as cell division proceeds. The model is simply defined by the rate of decrease of proliferative activity and the cell cycle duration. It is simple enough to be included in a virtual fruit. Future modelling should focus on the effect of environmental factors such as temperature, and plant factors such as carbon supply, on cell division intensity. A possible way would be to consider that cells divide when they double their initial size (Morgan et al., 2004; Yang et al., 2006) and thus to relate the intensity of proliferative activity to cell growth that is itself sensitive to temperature and carbon supply.
Cell differentiation:
Although the control of cellular differentiation has been investigated from a theoretical viewpoint (Cinquin and Demongeot, 2005), realistic and mechanistic models useful in the framework of a virtual fruit seem out of reach. There is clearly a need for models able to describe the phenomenology of cell differentiation as there is for cell division.
Cell size:
Cell size increase is driven by two distinct processes (Sugimoto-Shirasu and Roberts, 2003): cell growth, which involves the increase in total cytoplasmic macromolecular mass; and cell expansion, which involves the increase of cell volume through vacuolization.
It is stated that the amount of cytoplasm a cell can make and sustain is proportional to the amount of DNA in the nucleus (Sugimoto-Shirasu and Roberts, 2003). Amplifying genome size by endoreduplication contributes to increasing cell size (Kondorosi et al., 2000; Cheniclet et al., 2005). Although accumulating data revealed that endoreduplication is developmentally regulated (Joubès and Chevalier, 2000), it is still too poorly understood in plants to be formalized into a mechanistic model. That is why a phenomenological model of cell endoreduplication was developed in fruits (Bertin et al., 2006). The cells, which stopped mitosis, are assumed to be still able to perform an incomplete cycle, including DNA reduplication, but not cell division. The model considers two types of non-proliferating cells: those which become completely inactive and those which participate in incomplete cycles. The latter cells are distributed over several DNA levels according to the number of incomplete cycles they performed: the cells which participated in k incomplete cycles, increased their DNA level 2k times. Such a model is able to simulate the evolution of endoreduplication during fruit development. It has been shown that endoreduplication varies with environmental factors such as light, temperature, and nutrition (Barow, 2006). Further modelling work could consider such environmental effects.
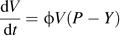
Assuming that the fruit mesocarp behaves as a single large cell, the Lockhart equation has been used by Fishman and Génard (1998) to model its volume increase. There is recent evidence that fruit growth is not always regulated by pressure changes in the fruit (Mingo et al., 2003). Lechaudel et al. (2007) proposed that ϕ and Y varied during fruit development, and Mingo et al. (2003) described a non-hydraulic regulation in cell wall extensibility. More research needs to be undertaken on the variation and control of these parameters. Although plastic variations of cell volume are the main determinants of cell expansion, the elastic variations of cell volume due to turgor pressure changes may also be important; they explain the diurnal tissue shrinkage, which has strong effects on the mineral accumulation in the fruit (Lang and Volz, 1998) and plays an important role in the control of physiological disorders. Therefore, a virtual fruit should combine plastic and elastic deformations. A first attempt has recently been made by Léchaudel et al. (2007) who showed by simulation the strong effect of the elastic modulus on mango fruit shrinkage. More knowledge is now needed on the mechanical properties of the cells and on their variation.
An important question emerging from the above considerations is how to link cell growth due to endoreduplication and expansion. It may be assumed that the rate of carbohydrate uptake by cells is proportional to their level of endoreduplication, but more quantitative research is needed to test such an hypothesis.
Metabolic transformations and vacuolar storage:
Taste mainly results from the accumulation of sugars and acids in fruit cells (Stevens et al., 1979). This accumulation can be controlled through the intensity of metabolic transformations and/or through the vacuolar storage capacity. These processes are well known and have been extensively described in the literature (Ho, 1988; Wink, 1993). On this basis, Génard and Souty (1996) and Génard et al. (2003) designed a mechanistic model called SUGAR to predict the changes in sugar composition during peach fruit development. In this model, the unloaded sugars are either directly stored in the cells, transformed into other sugars, or used to synthesize other compounds (structural carbohydrates, etc). Lobit et al. (2003, 2006) designed two models predicting fruit acidity. The first one described citric acid production and degradation during fruit development by representing the fluxes through the citrate cycle. In the second one, malic acid content was assumed to depend mainly on the cell's capacity to store this acid in its vacuole, i.e. the thermodynamic conditions of the transport from the cytosol to the vacuole. Both citric and malic models were combined with a model of pH calculation (Lobit et al., 2002) into a global model able to predict the titratable acidity of the fruit (Habib, 2000).
Compounds from secondary metabolism such as vitamins and carotenoids are essential for the nutritional quality of fruits. Their biosynthetic pathways are often known (Carrari and Fernie, 2006), but the lack of knowledge on their regulation strongly limits modelling capacity in the framework of a virtual fruit. It is known that environmental factors and agricultural techniques affect secondary compounds (Dumas et al., 2003; Gautier et al., 2005a), but more quantitative studies are needed before undertaking modelling. There is a real challenge for the future here. As the biosynthesis of these compounds is partly regulated by ethylene (Marty et al., 2005) and because this hormone is strongly related to maturation and growth duration in climacteric fruits, ethylene production has to be considered in a virtual fruit. Génard and Gouble (2005) have recently proposed a model in which the biosynthetic pathway of ethylene is supplied by ATP and regulated by 1-aminocyclopropane-1-carboxylic acid synthase and 1-aminocyclopropane-1-carboxylic acid oxidase. The respiration, which is an essential variable of this model, has been modelled according to the concept of growth and maintenance respiration (Thornley, 1970), but a more physiologically based representation of respiration could be used in the future (Cannel and Thornley, 2000; Dewar, 2000).
Although the theories underlying these models are largely different from one another, the same general laws have been used for modelling. In particular, the enzymatic reactions have been described according to the ‘rate law’ of chemical kinetics (Chang, 2000), which states that the rate of a reaction is proportional to the reactant. This conceptual homogeneity should favour the integration of the different models into a virtual fruit. Most of the processes involved at the cell scale have been described through published models and can be assembled as shown in Fig. 2.
Tissue scale
In a virtual fruit focusing on fruit quality, each of the fruit tissues would be intrinsically characterized by its quality criteria (e.g. sweetness, acidity, and nutritional quality would be considered only in the edible tissues such as pericarp; size would be considered for every tissue) and the related important processes. The process intensity should be tissue specific. For example, growth respiration is known to be higher for lignified tissues than for other tissues (Williams et al., 1987) so the parameters of growth respiration models presented at the cell scale need to be tissue specific. In tomato fruit which has been intensively studied, it is now well accepted that the metabolism in the fruit pericarp is different from that in the placenta, and this is even different from that in the columella (Carrari and Fernie, 2006). It has been also shown that the composition of secondary compounds varied in skin, flesh, and seeds of tomatoes (Toor and Savage, 2005). Similarly, biophysical characteristics such as elasticity, plasticity, and conductance to water are tissue specific. In a mechanistic approach, Bussières (1994) proposed a model of water import in tomato fruit in which conductances of the mesocarp and placenta vessels may be different. In the case of the skin, these biophysical characteristics are especially important because they have strong effects on fruit growth (Thompson et al., 1998; Andrews et al., 2000) and on skin cracking (Gibert et al., 2005). More basic research is needed on the variations of composition and biophysical characteristics of the tissues.
Another important point is that the tissue cannot be considered as being a simple juxtaposition of independent cells. Indeed, Bertin (2005) showed that there was a clear linear decrease of cell size with the increase of cell number in tomato pericarp, which indicates a density dependence effect. She also found that pericarp mass increases with increasing cell number in spite of competition, which indicates an undercompensating density dependence. It is clear that competition between cells has to be taken into account in a virtual fruit. One important question for the future is to define how to represent this competition. Lescourret and Génard (2003) proposed a multilevel theory of competition between fruit units which could be adapted to the case of competition among cells. Another important question is the impact of cell competition on cell metabolism and thus on tissue composition. Cell competition also potentially affects the biophysical characteristics of the tissue (e.g. the cell size has an effect on tissue texture; Sams, 1999). There is clearly a need for research on these different points and on how to model the effect of cell competition on cell growth, composition, and biophysics.
Fruit scale
Important quality traits are manifested at the fruit scale. This is especially true for fruit size, dry matter content, and percentage of edible tissues. Fruit size and dry matter content result from the exchange of resources with the plant and the atmosphere. The carbohydrate supply has often been modelled according to the source/sink concepts (Baumgaertner et al., 1984; Gutierrez et al., 1985; Gary et al., 1998, Léchaudel et al., 2005). An important variable of these models is the fruit demand for carbohydrates, which is positively correlated to the seed number (Lescourret et al., 1998). In some models, a more mechanistic approach has considered the processes involved in sugar unloading from the phloem to the fruit tissues. For example, Fishman and Génard (1998) and Bruchou and Génard (1999) modelled sugar phloem unloading through mass flow, diffusion, and active transport. This approach should be adopted in the virtual fruit to relate water and carbohydrate fluxes mechanistically. The phloem unloading pathway (symplasmic or apoplasmic) is an important aspect of the regulation of sugar unloading, with a possible shift from a symplasmic to apoplasmic pathway during fruit development, as shown for grape berry (Zhang et al., 2006). More modelling effort is needed on this shift. The fluxes of water in xylem and phloem tissue have been extensively modelled at the scale of the vessel (Tyree et al., 1974) or the plant (Boersma et al., 1991; Doussan et al., 1998), but only a few fruit models describe them (Bussières, 1994, 1995; Fishman and Génard, 1998). The differences in water potential, osmotic potential, and turgor pressure between the stem and the fruit have been considered as the driving forces of the water import rate, and the role of pedicel resistance and variation in xylem functionality has been emphasized recently (Bussières, 2002; Drazeta et al., 2004). The water balance of the fruit has been calculated considering water uptake and transpiration per unit of fruit area as constant (Lee, 1990) or variable (Bussières, 1993; Génard and Huguet, 1996; Fishman and Génard, 1998; Léchaudel et al., 2004). These works constitute a basis for modelling the water balance of a virtual fruit. The xylem ‘backflow’ from the fruit to the plant is an important aspect of fruit water balance which is now well known (Lang and Thorpe, 1989; Keller et al., 2006), although it still requires a modelling effort.
In a postharvest context, the gas (O2, CO2, ethylene, and water vapour) fluxes through the skin have been related to concentration gradients, according to physical laws of gas diffusion (Ben-Yehoshua and Cameron, 1989). This physical approach could be adequate in the framework of a virtual fruit. A model of fruit surface conductance to water vapour has been proposed recently (Gibert et al., 2005) which could improve the prediction of fruit transpiration as a function of fruit growth. More generally, fruit surface conductance to gas needs to be modelled in the future because it has a strong implication for fruit quality through its effect on fruit physiology and ripening (Paul and Srivastava, 2006).
Combining the model of carbon and water balance proposed by Fishman and Génard (1998), the model of Bussières (2002) on pedicel resistance, and that of Gibert et al. (2005) on fruit surface conductance to water vapour would provide a sound basis to model the water and carbon exchanges with the plant and the atmosphere.
Biomass allocation to the fruit tissues (which determines the percentage of edible tissues) also needs to be considered at the fruit scale. However, to our knowledge, there is no mechanistic model of biomass allocation into fruit tissues. Establishing empirical laws relating the size of any given tissue to that of another one or to that of the whole fruit may be a solution. Such laws are common in biology, mainly in the framework of allometric growth (Causton and Venus, 1981; West et al., 1997). For example, the dry masses of each fruit tissue and the dry mass of the fruit have been linked by allometric relationships (Léchaudel et al., 2002; Lescourret and Génard, 2005), which seem to be independent of assimilate supply, but vary greatly according to the genotype (Quilot et al., 2004). Derivation of allometric equations could be used in a virtual fruit to distribute the carbon unloaded to the fruit between its different tissues as done by Lescourret and Génard (2005).
Which controls for a virtual fruit?
Fruit development is controlled by three classes of factors: environmental, genetic, and plant (Fig. 1). The first class includes temperature, light, and air humidity. Such factors influence important processes such as cell cycle duration (Francis and Barlow, 1988), photosynthesis and respiration (Pavel and Dejong, 1993), transpiration (Leonardi et al., 1999), phloemic transport (Guichard et al., 2005), and metabolism (Yamada, 1994). The influence of the main environmental factors on plant physiology has classically been included in most ecophysiological models. Except for temperature effects (Bussières, 1995, 2002), this has been done more rarely for fruit, though its microclimate strongly affects its quality (Gautier et al., 2005b). Recent advances on the climate perceived by individual plant organs (Chelle, 2005) suggest that fruit temperature models will soon be available for coupling with a virtual fruit. A large body of literature shows the importance of genetic factors in the control of fruit quality (Dirlewanger et al., 1999; Causse et al., 2002). Genetic control has recently been integrated in some ecophysiological models (Yin et al., 2004) including fruit models (Quilot et al., 2004). Considering that only a limited number of genes present in the fruit are involved, or that groups of genes have co-ordinated actions (Tardieu, 2003), the genetic control could be defined by a limited set of genotype-specific parameters of the main modelled processes (a few tens to hundreds, compared with the thousands of genes controlling fruit development). A first attempt at genotypic specification for a fruit model has been made by looking for quantitative trait loci (QTLs) of parameters (Quilot et al., 2005b). With the rapid progress of functional genomics, a further step would be, according to Struik et al. (2005), to combine fruit models with genes and gene regulatory networks underlying these QTLs for key subprocesses controlling fruit quality traits.
The plant controls fruit quality through resource and hormonal controls. Source–sink relationships for carbon in plants and their effect on fruit growth (Starck et al., 1990; Ho, 1992, 1996) and fruit metabolism (Archbold, 1999; Souty et al., 1999) are well documented. Water relations in plants and their effects on fruit quality have also been intensively studied (Ho et al., 1987; Mitchell et al., 1991). Carbon and water allocation within a plant depends on complex rules linking source organs (leaves for carbon and roots for water) and sink organs such as fruits. The complexity essentially comes from regulations due to feedback mechanisms and interactions among different functions (e.g. between sink growth and leaf photosynthesis). Plant models of carbon and water allocation able to simulate this complexity have been developed during the last 30 years (Le Roux et al., 2001; Van Ittersum et al., 2003), with a recent focus on 3D virtual plants (Dauzat et al., 2001; King, 2005). These models are powerful tools to analyse how the plant affects the carbon and water accumulation in fruit.
The phloem sap sugar concentration and water potential in the plant would be the main plant variables controlling a virtual fruit. The virtual fruit will need to be connected to a plant model of carbon and water balance able to predict these two variables. Plant hormonal control of fruit quality is very difficult to take into account even if it has been extensively investigated. More quantitative research is needed in this field.
The virtual fruit as a tool for agronomists and geneticists
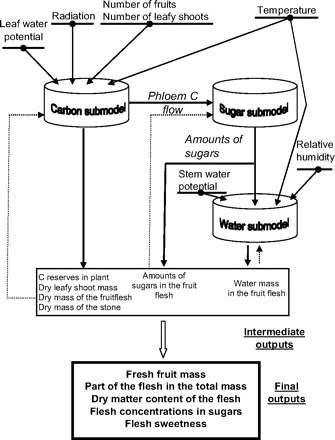
Using a virtual fruit in conjunction with models of plant functioning would make it possible to simulate the effects of agronomical factors, such as water availability, on fruit quality. This has been illustrated in the first version of the virtual fruit by Lescourret and Génard (2005). This first virtual fruit was developed by adapting and integrating into one complex system three existing process-based models describing fruit dry mass growth (Lescourret et al., 1998), sugar accumulation in the flesh (Génard et al., 2003), and fruit fresh mass growth (Fishman and Génard, 1998) by supply–demand, compartmental, and biophysical approaches, respectively. A general scheme of this first virtual fruit is given in Fig. 3.
General scheme of the Lescourret and Génard (2005) virtual fruit. Underlined variables are external variables, arrows are data flows, and dashed lines represent feedback information. (This figure has been redrawn from Lescourret F, Génard M. 2005. A virtual peach fruit model simulating changes in fruit quality during the final stage of fruit growth. Tree Physiology25, 1303–1315, and reproduced by kind permission of Heron Publishing.)
The model of Lescourret et al. (1998) describes the carbon balance of a fruit-bearing stem. The daily available pool of carbon assimilates consists of leaf assimilation plus, eventually, the carbon mobilized from reserves. The leaf photosynthesis may be affected by a feedback inhibition through the size of leaf reserves. Carbon is allocated according to organ demands and priority rules. Maintenance respiration costs have first priority, whereas vegetative and reproductive growth are given second and third priorities, respectively. The carbon demand for fruit growth emphasizes the role of fruit history, in terms of both sink size and sink activity. It also emphasizes the role of developmental time. The incoming carbon flow is shared between the flesh and the endocarp+seed according to an equation derived from an empirical relationship between endocarp+seed dry mass and total fruit dry mass. Using the model of Génard et al. (2003), the fruit flesh carbon is then partitioned into several compounds: four sugars (sucrose, sorbitol, glucose, and fructose), other fruit compounds globally considered (starch and structural carbohydrates), and respired CO2. The rates of change of the amounts of carbon in the four sugar compounds are described through a set of differential equations. According to the Fishman and Génard (1998) model, the flow of water to the fruit is driven by differences in hydrostatic and osmotic pressures between xylem or phloem and fruit. The fruit osmotic pressure induced by sugars is calculated by means of the ‘sugar accumulation’ model. The hydrostatic pressure is calculated assuming that the growth predicted by the Lockhart equation is equal to the water inflows minus fruit transpiration. Fruit transpiration is calculated from skin conductance to water vapour and the vapour pressure difference between the air and the fruit.
The inputs of this first virtual fruit are weather data (i.e. global radiation, temperature, and air relative humidity), stem water potential, and leafy shoot and fruit number on the stem (Fig. 3). Although each of the three initial submodels has already been validated, the virtual fruit was evaluated as a whole. It was successfully done for several quality traits (fruit fresh mass, proportion of flesh in the total mass, dry matter content of flesh, sucrose, sorbitol, glucose, and fructose concentrations in the flesh, and sweetness index) on peach fruit using experiments from three different years. The relative root mean squared error of prediction calculated using a cross-validation approach (Wallach et al., 2001) was smaller than 20% in most cases (Lescourret and Génard, 2005). Moreover, a simplified version (for sugars only) was tested on 87 different peach genotypes, and the error of prediction was also smaller than 20% in most cases (Quilot et al., 2005a).
Virtual experiments using this model showed that applying a water stress to peach trees after a period of normal water supply resulted in an intense slowing down of growth. During the same period, the fruits grown under continuous water stress experienced continuous growth. This suggests that continuously stressed fruit may adapt to drought. In the virtual fruit, this adaptation was based on a ‘sugar signal’, i.e. the increase of sugar concentration during the stress period being able to promote the growth. Thus it appears that a virtual fruit can develop complex adaptations. Combining a virtual fruit with a virtual plant could give rise to new tools for agronomists in charge of technical innovations to be able to improve fruit quality. Virtual experiments in which several agronomic factors could be combined would make it possible to find the best technical combinations in terms of fruit quality.
Geneticists have made much progress in the last decade thanks to molecular marker technologies, which enable dissection of the variation of traits into effects of QTLs. However, they still encounter two major difficulties. First, the quality traits that are breeding targets result from many overlapping processes and are thus controlled by many genes. The second difficulty results from the fact that these characters are under the influence of the environment. This often results in strong genotype×environment interactions which make the genetic analysis and its application to breeding difficult. Studying a quantitative trait via a virtual fruit makes it possible to deal with both difficulties simultaneously. Instead of directly looking for QTLs controlling this trait, it is more efficient to look for QTLs controlling model parameters because they are assumed to be independent of the environment. This approach was applied by Quilot et al. (2005a, b) to peach fruit. Through the study of the co-locations between QTLs of parameters and traits, physiological meanings of QTLs of traits have been proposed. For instance, on the first linkage group (LG1), QTLs for fresh fruit mass are located in the same region as QTLs for parameters involved in sugar metabolism and early fruit growth. On LG2, LG4, and LG7, they are co-located with QTLs for parameters involved in water fluxes in the fruit (hydraulic conductance and permeation coefficient of the fruit surface to water vapour). Quilot et al. (2005a, b) also observed pleiotropic effects. For instance, QTLs for parameters involved in sugar metabolism were located in the same region as a QTL for total sugar concentration on LG1 and in the same region as a QTL for dry matter content of flesh on LG3.
Moreover, when parameters are well predicted by QTLs, it is then possible to predict, through the virtual fruit, the quality traits (i.e. fruit mass) of any given genotype in a given environment. Assuming for instance that one QTL could be manipulated to increase the intensity of sucrose uptake by the fruit, the Lescourrret and Génard (2005) ‘Virtual Fruit’ model predicts an increase of both carbon and water stored in the fruit and thus an increase of fruit size (Fig. 4). It also predicts an increase in fruit sugar concentration and leaf photosynthesis, and a decrease in the leaf carbohydrates reserve pool (Fig. 3). Indeed, the change in assimilate uptake by the fruit alters not only the fruit function but also the source–sink relationship within the plant. This example shows that a virtual fruit can be a powerful tool for analysing the response of genotypes, in terms of fruit quality and plant functioning, to contrasting environmental conditions and to look for the genotype adaptation. This opens up the way to new breeding approaches based on simulation results.
The Lescourret and Génard (2005) virtual fruit model was used to analyse the effect of a genetic change in mean sugar uptake by the peach fruit on the dynamics of its quality [fruit mass and sugar concentration (g 100 g−1 fresh mass), carbohydrate reserve concentrations in the leaves (g 100 g−1 dry mass), and leaf photosynthesis] from 81 d to 139 d after full bloom. That model is a combination of a ‘carbon submodel’, describing the management of carbon in the fruit-bearing shoot and the growth in dry mass of the vegetative and fruit parts, a ‘sugar submodel’ that partitions the carbon into the four sugars present in the peach fruit flesh, and a ‘water submodel’ describing the management of water in the fruit.
Conclusion
Fruit quality traits are quantitative, complex, and controlled by environment and genes. The variation of these traits with environment and genotype has been traditionally studied following a quantitative genetics approach such as correlation and marker–trait association analysis. The combined physiological and genetic approach of our virtual fruit research is more able to take into consideration the internal regulations and their genetic and environmental controls. The application of the first version of a virtual fruit to agronomic and genetic questions is encouraging and is an important step towards quality trait modelling of fruits using different biological scales for explanation (Struik et al., 2005).
Gutiérrez et al. (2005) proposed a systems approach to understand plant biology from the molecular to the ecological scale. This systems biology is based on the integration of existing knowledge about biological components and building of the system as a whole. In accordance with this viewpoint, Yin et al. (2004) proposed that a new generation of models should enable us to narrow the gap between genes and complex phenotypes. Concerning fruit quality, this new generation is really needed to accompany the advances in fruit genomics (Baxter et al., 2005). We believe that a virtual fruit able to generate complex phenotypes thanks to its emerging properties belongs to this new generation.
Abbreviation
We thank G Wagman and A Hall for revising the manuscript.



![The Lescourret and Génard (2005) virtual fruit model was used to analyse the effect of a genetic change in mean sugar uptake by the peach fruit on the dynamics of its quality [fruit mass and sugar concentration (g 100 g−1 fresh mass), carbohydrate reserve concentrations in the leaves (g 100 g−1 dry mass), and leaf photosynthesis] from 81 d to 139 d after full bloom. That model is a combination of a ‘carbon submodel’, describing the management of carbon in the fruit-bearing shoot and the growth in dry mass of the vegetative and fruit parts, a ‘sugar submodel’ that partitions the carbon into the four sugars present in the peach fruit flesh, and a ‘water submodel’ describing the management of water in the fruit.](https://oup.silverchair-cdn.com/oup/backfile/Content_public/Journal/jxb/58/5/10.1093/jxb/erl287/2/m_jexboterl287f04_lw.gif?Expires=1716474971&Signature=QbudQG9lAKZvPLhhjSSFcSBlLERzff4VeStkrVR9eh-0ykObUry-f-hN86DYSZSEtfY9~DvX6tWfW1j9sO~iII3AAqmHuC69cVNGzFPfMmSM-R0CQvrYOHN54K3dXYo48NBlH1~fJqQGmDg9P8RwlKaKlNKkvi6QWDlJI-eKQt3Cck4~TiGP0mEYXZSQgndlPUuABkjfxbqpAQ7LoMAIIY8TCZ8hkTnvGUzeC43sBE16sw-mWy-4FjpanrOc7TteiJlE0deu0nomu817oer8Ru7TUZtZjpoi5bA9swCkiGP9D6~7cG4POzE7EVnCNwR4~yOXdvj2344Pg9qleDnqKA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

Comments